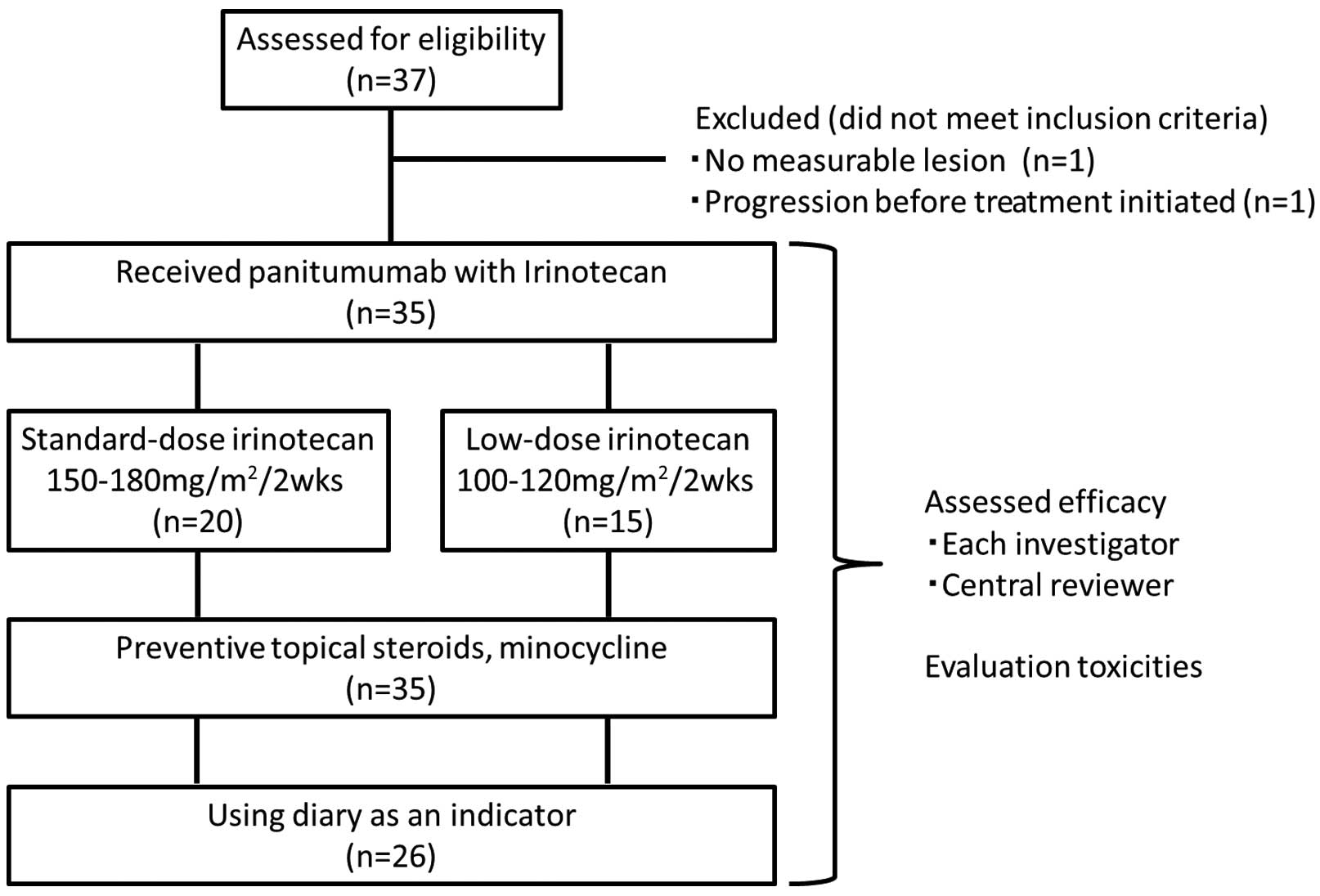
Phase II trial of panitumumab with irinotecan as salvage therapy for patients with advanced or recurrent colorectal cancer (TOPIC study)
![Pharmacokinetics, Metabolism, and Excretion of Irinotecan (CPT-11) Following I.V. Infusion of [14C]CPT-11 in Cancer Patients | Drug Metabolism & Disposition Pharmacokinetics, Metabolism, and Excretion of Irinotecan (CPT-11) Following I.V. Infusion of [14C]CPT-11 in Cancer Patients | Drug Metabolism & Disposition](https://dmd.aspetjournals.org/content/dmd/28/4/423/F1.large.jpg)
Pharmacokinetics, Metabolism, and Excretion of Irinotecan (CPT-11) Following I.V. Infusion of [14C]CPT-11 in Cancer Patients | Drug Metabolism & Disposition
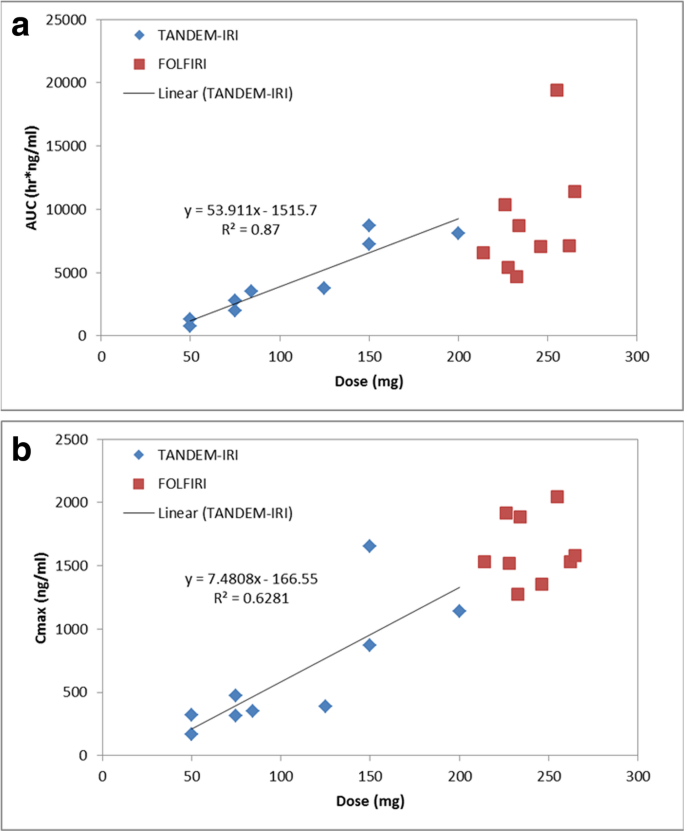
Selective TACE with irinotecan-loaded 40 μm microspheres and FOLFIRI for colorectal liver metastases: phase I dose escalation pharmacokinetic study | BMC Cancer | Full Text

Cost–effectiveness of UGT1A1 genotyping in second-line, high-dose, once every 3 weeks irinotecan monotherapy treatment of colorectal cancer | Pharmacogenomics
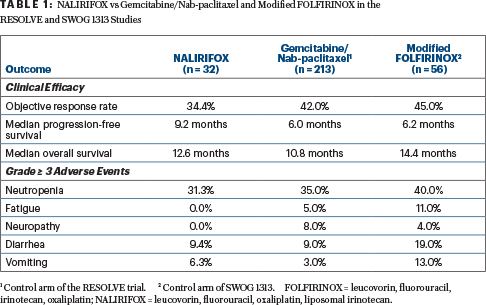
First-Line Liposomal Irinotecan–Containing Regimen Studied in Advanced Pancreatic Cancer - The ASCO Post

Liposomal irinotecan (Onivyde): Exemplifying the benefits of nanotherapeutic drugs - Milano - 2022 - Cancer Science - Wiley Online Library

DEP® irinotecan synergistic with Lynparza® in refractory human colon cancer model | BioMelbourne Network

JCM | Free Full-Text | Safety, Feasibility and Technical Considerations from a Prospective, Observational Study—CIREL: Irinotecan-TACE for CRLM in 152 Patients

UGT1A1*28 genotype and irinotecan dosage in patients with metastatic colorectal cancer: a Dutch Colorectal Cancer Group study | British Journal of Cancer

Modified XELIRI (capecitabine plus irinotecan) versus FOLFIRI (leucovorin, fluorouracil, and irinotecan), both either with or without bevacizumab, as second-line therapy for metastatic colorectal cancer (AXEPT): a multicentre, open-label, randomised ...