
Immune checkpoint inhibitor-based combinations: is dose escalation mandatory for phase I trials? - Annals of Oncology

Bayesian Adaptive Design for Finding the Maximum Tolerated Sequence of Doses in Multicycle Dose-Finding Clinical Trials | JCO Precision Oncology
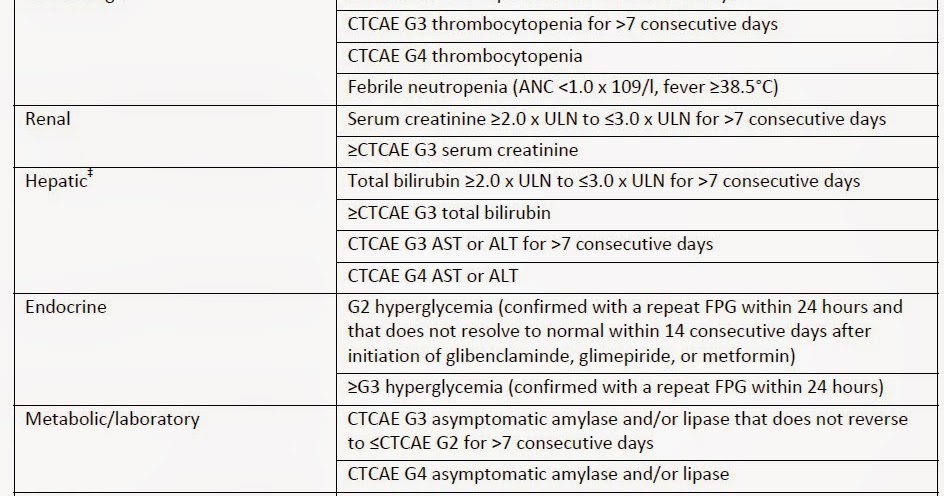
On Biostatistics and Clinical Trials: Dose Limiting Toxicity (DLT) and Common Toxicity Criteria (CTC) / Common Terminology Criteria for Adverse Events (CTCAE)

Towards new methods for the determination of dose limiting toxicities and the assessment of the recommended dose for further studies of molecularly targeted agents – Dose-Limiting Toxicity and Toxicity Assessment Recommendation Group
A phase I study of escalated dose subcutaneous alemtuzumab given weekly with rituximab in relapsed chronic lymphocytic leukemia/small lymphocytic lymphoma | Haematologica

Heterogeneity in the definition of dose-limiting toxicity in phase I cancer clinical trials of molecularly targeted agents: a review of the literature. | Semantic Scholar

Early-drug development in the era of immuno-oncology: are we ready to face the challenges? - Annals of Oncology

Illustration of the chronic dose-limiting toxicity (DLT) concept. (*)... | Download Scientific Diagram
Dose Limiting Toxicities (DLT) DOSE LIMITING TOXICITIES (DLT) Example: Dose escalation will proceed within each cohort according

ELIZABETH GARRETT-MAYER (SOME SLIDES BY PAT LORUSSO OF KARMANOS CANCER INSTITUTE WAYNE STATE UNIVERSITY) Phase I Trials of Chemotherapy and Targeted Agents. - ppt download

Towards new methods for the determination of dose limiting toxicities and the assessment of the recommended dose for further studies of molecularly targeted agents – Dose-Limiting Toxicity and Toxicity Assessment Recommendation Group











