Phase I dose-escalation study to determine the safety, tolerability, preliminary efficacy and pharmacokinetics of an intratumoral injection of tigilanol tiglate (EBC-46) - eBioMedicine

Innovative design for a phase 1 trial with intra-patient dose escalation: The Crotoxin study | Semantic Scholar
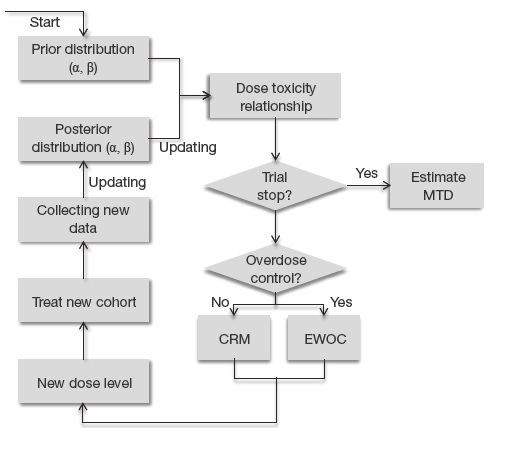
A new pragmatic design for dose escalation in phase 1 clinical trials using an adaptive continual reassessment method | BMC Cancer | Full Text

Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial - The Lancet

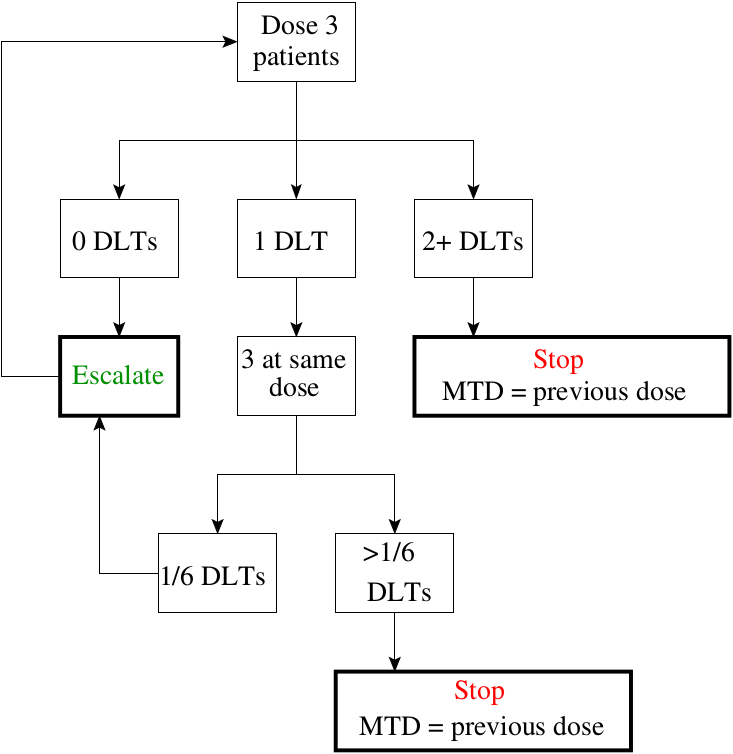
The 3 + 3 dose escalation study design. DLT, dose limiting toxicity;... | Download Scientific Diagram

توییتر \ Stephen V Liu, MD در توییتر: «#OncoAlert #AACR20 Dose escalation of TAK-788 (mobocertinib) established MTD of 160mg qday. DLTs were 1 case each of grade 3 diarrhea, grade 3 mucositis,

Contemporary dose-escalation methods for early phase studies in the immunotherapeutics era - ScienceDirect

Dose intra‐subject escalation to an event (DIETE): A new method for phase 1 dose‐finding utilizing systematic intra‐subject dose escalation with application to T‐cell engagers - Xu - 2021 - Pharmaceutical Statistics -

Contemporary dose-escalation methods for early phase studies in the immunotherapeutics era - European Journal of Cancer

Figure 1 | Phase I dose-escalation trial of the oral AKT inhibitor uprosertib in combination with the oral MEK1/MEK2 inhibitor trametinib in patients with solid tumors | SpringerLink

Ustekinumab dose escalation: An option for nonresponsive Crohn's? | Latest news for Doctors, Nurses and Pharmacists | Pharmacy

Innovative design for a phase 1 trial with intra-patient dose escalation: The Crotoxin study - ScienceDirect
Protocol B7861002 A PHASE 1 DOSE ESCALATION STUDY TO EVALUATE THE SAFETY, PHARMACOKINETICS AND PHARMACODYNAMICS OF INTRAVENOUS P
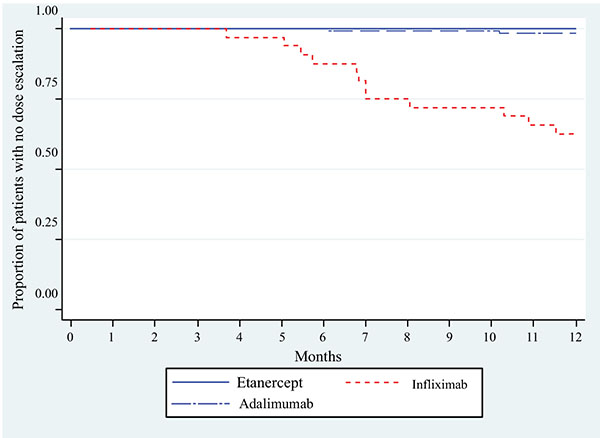
Dose Escalation and Co-therapy Intensification Between Etanercept, Adalimumab, and Infliximab: The CADURA Study
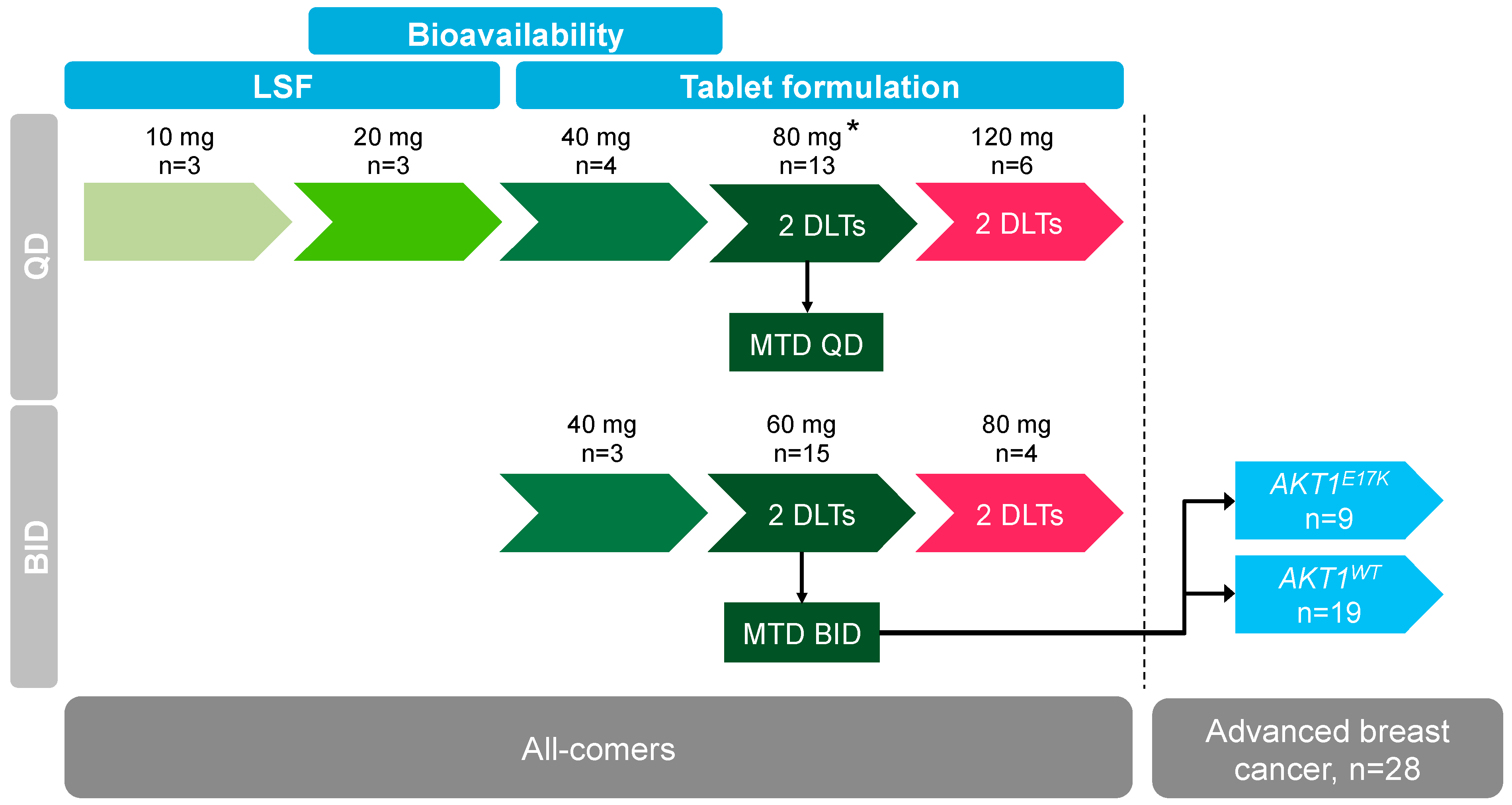
Cancers | Free Full-Text | Phase 1 Dose Escalation Study of the Allosteric AKT Inhibitor BAY 1125976 in Advanced Solid Cancer—Lack of Association between Activating AKT Mutation and AKT Inhibition-Derived Efficacy