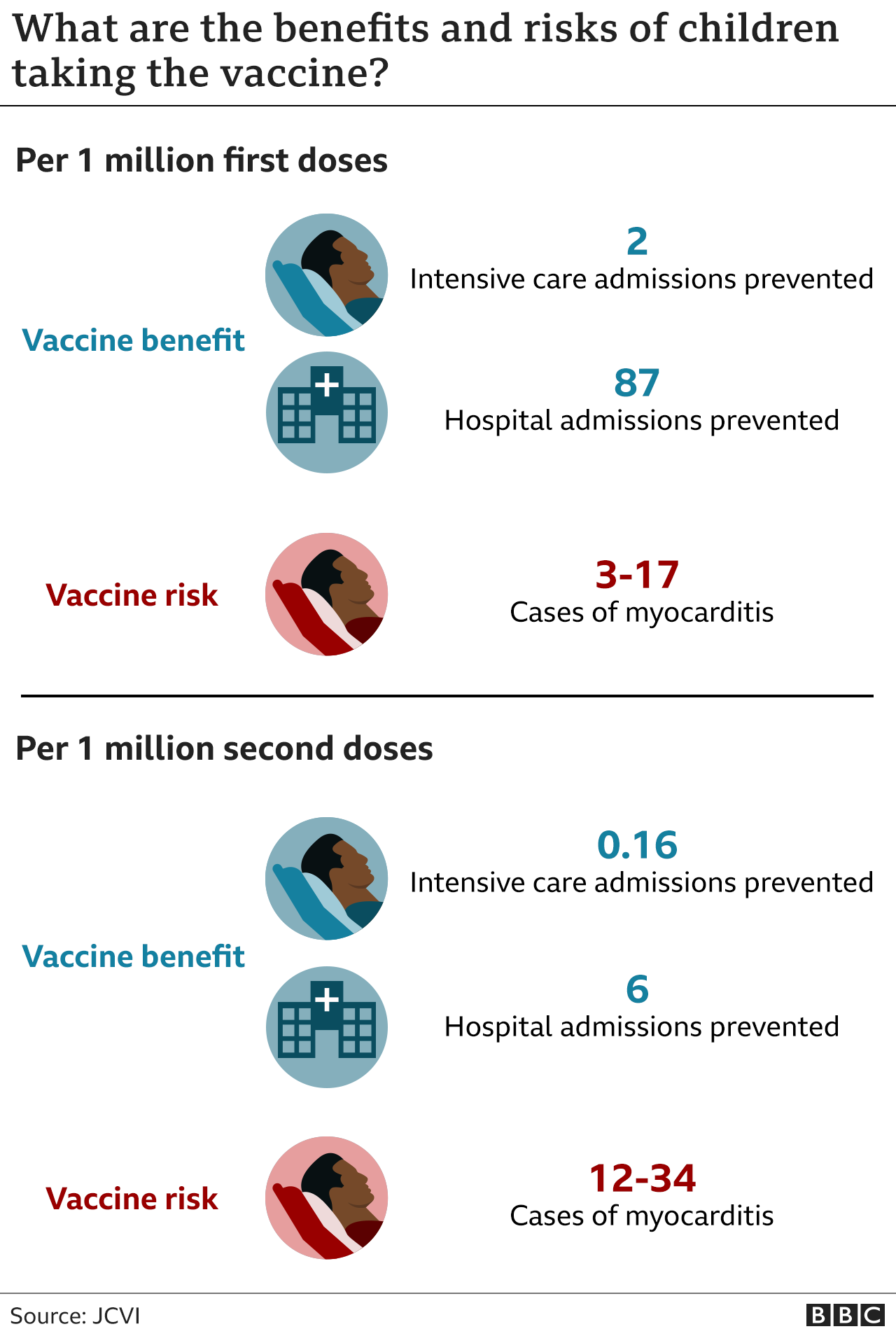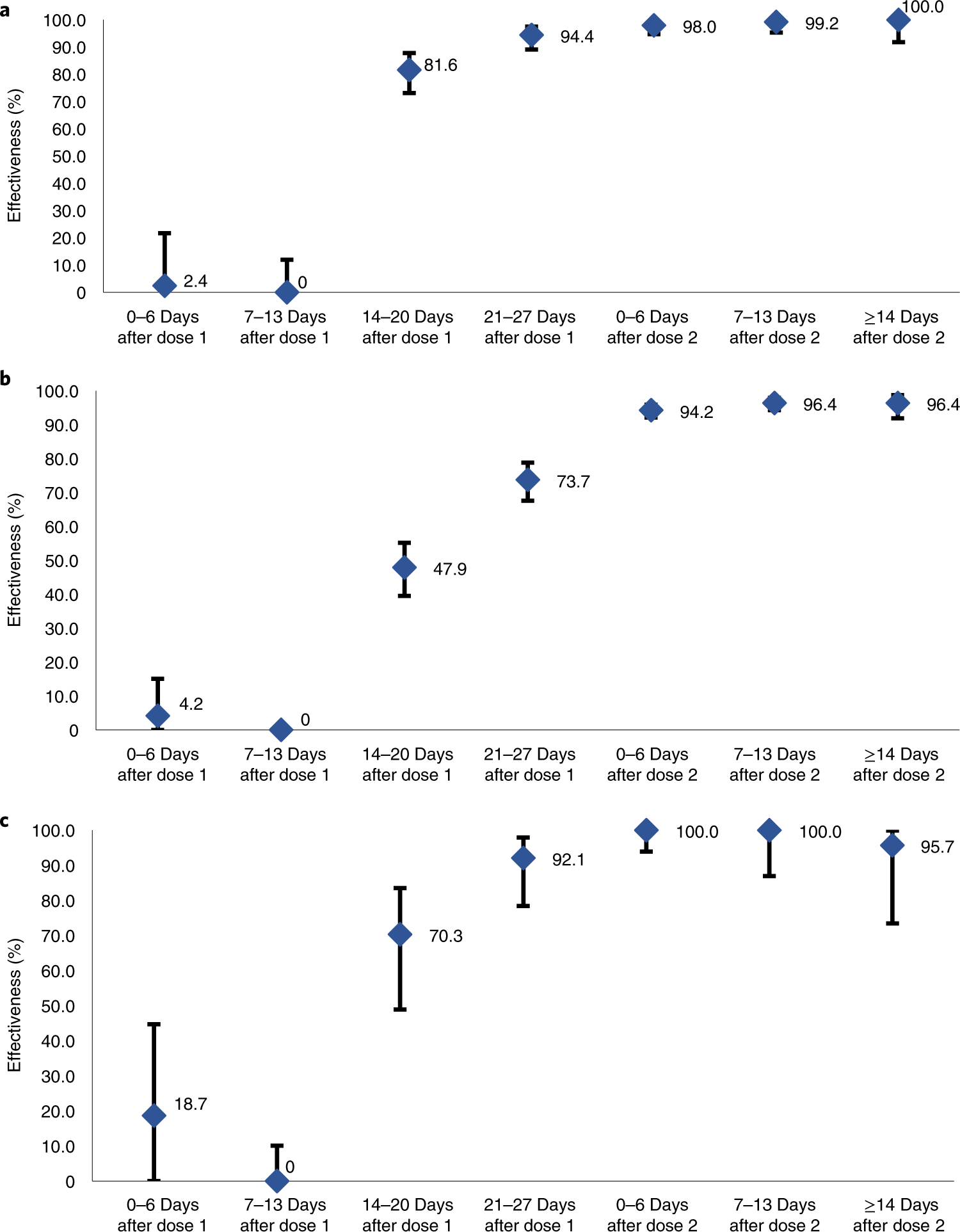
mRNA-1273 COVID-19 vaccine effectiveness against the B.1.1.7 and B.1.351 variants and severe COVID-19 disease in Qatar | Nature Medicine
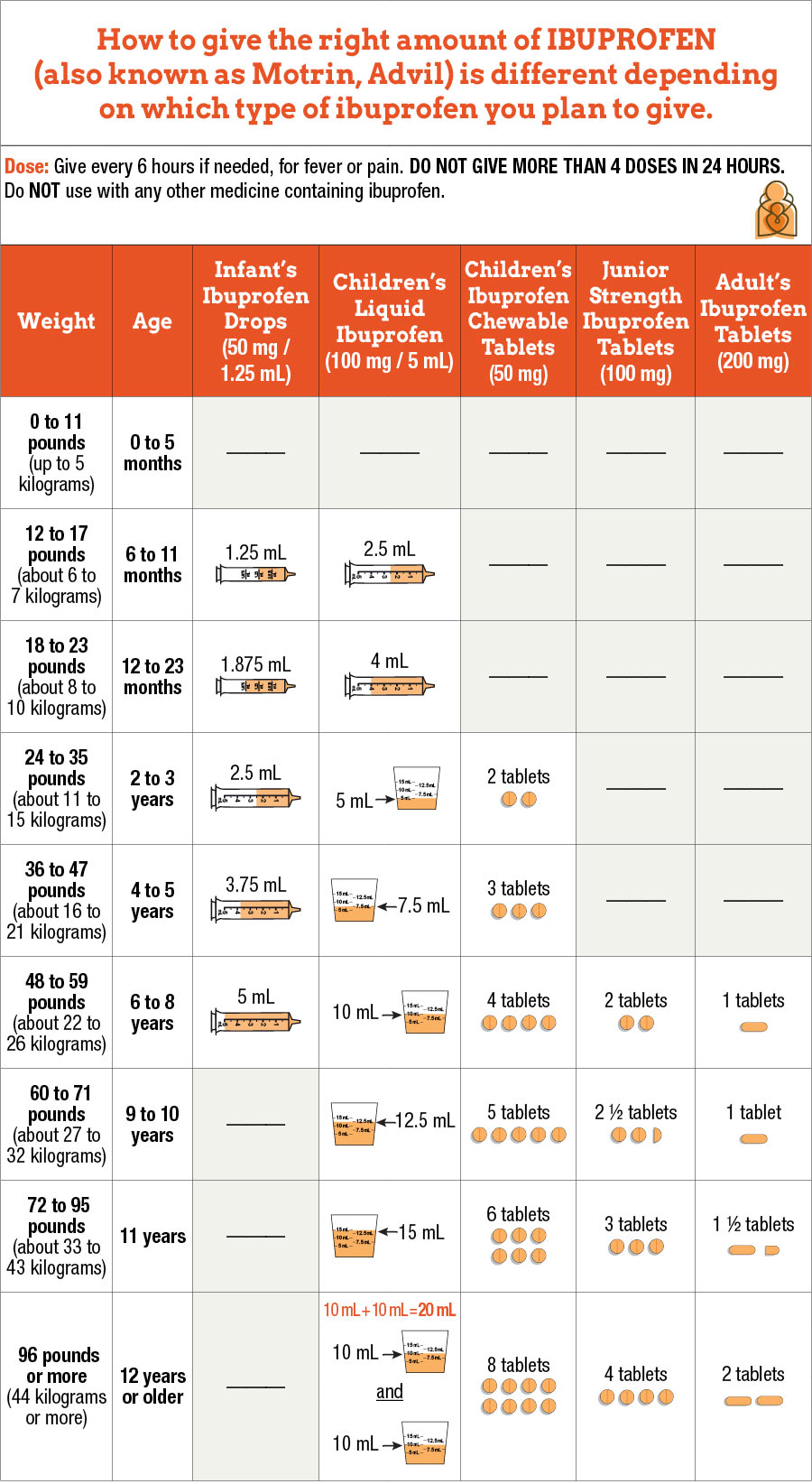
Should FDA Reconsider Allowing the Pooling of COVID-19 Vaccine Doses to Obtain Additional Doses? | Institute For Safe Medication Practices

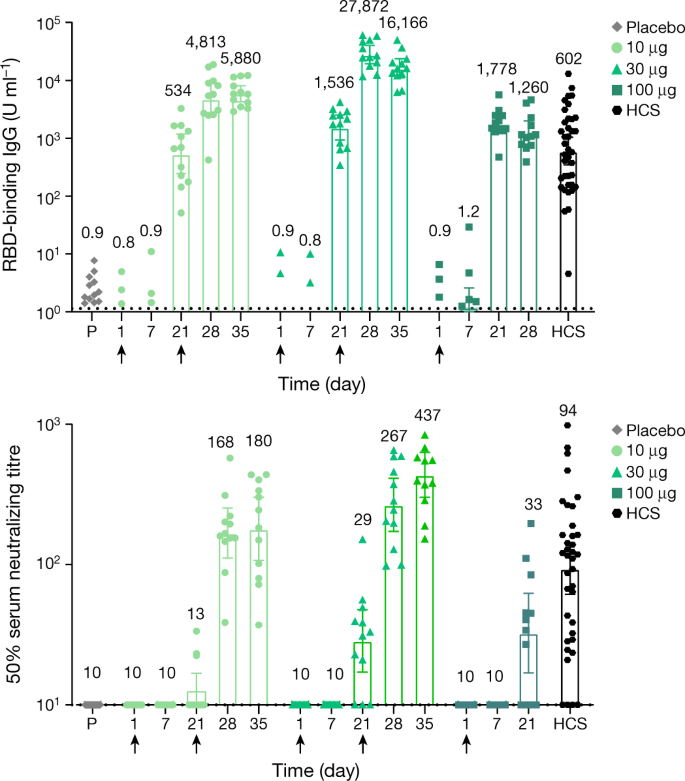
SARS-CoV-2 mRNA vaccine induces robust specific and cross-reactive IgG and unequal neutralizing antibodies in naive and previously infected people - ScienceDirect

I got AstraZeneca for my first dose. Which should I get for my second?' A COVID vaccination guide - Healthy Debate
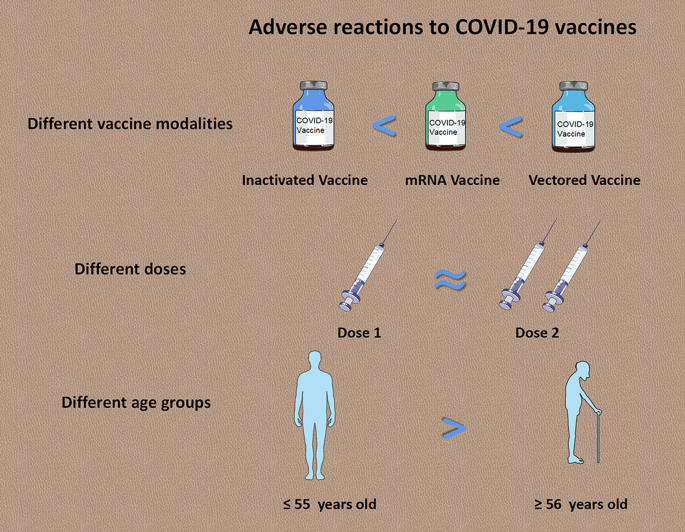
Safety of SARS-CoV-2 vaccines: a systematic review and meta-analysis of randomized controlled trials | Infectious Diseases of Poverty | Full Text

Effectiveness of a third dose of BNT162b2 mRNA COVID-19 vaccine in a large US health system: A retrospective cohort study - The Lancet Regional Health – Americas

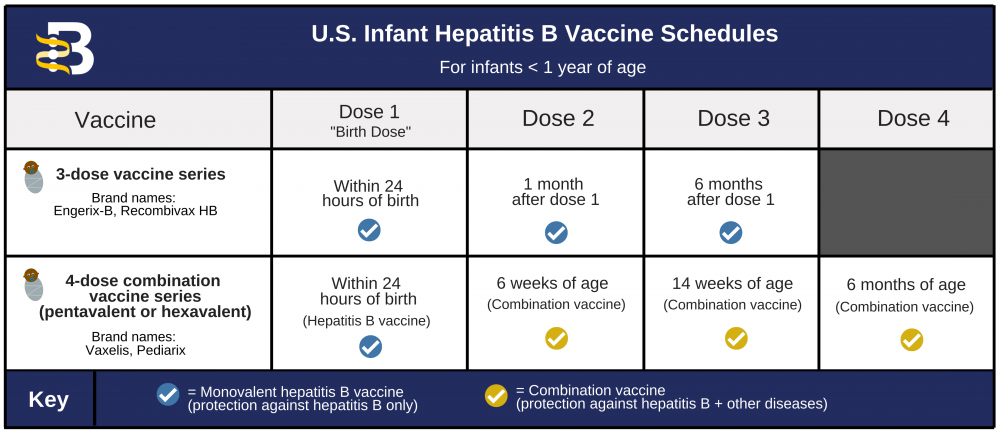
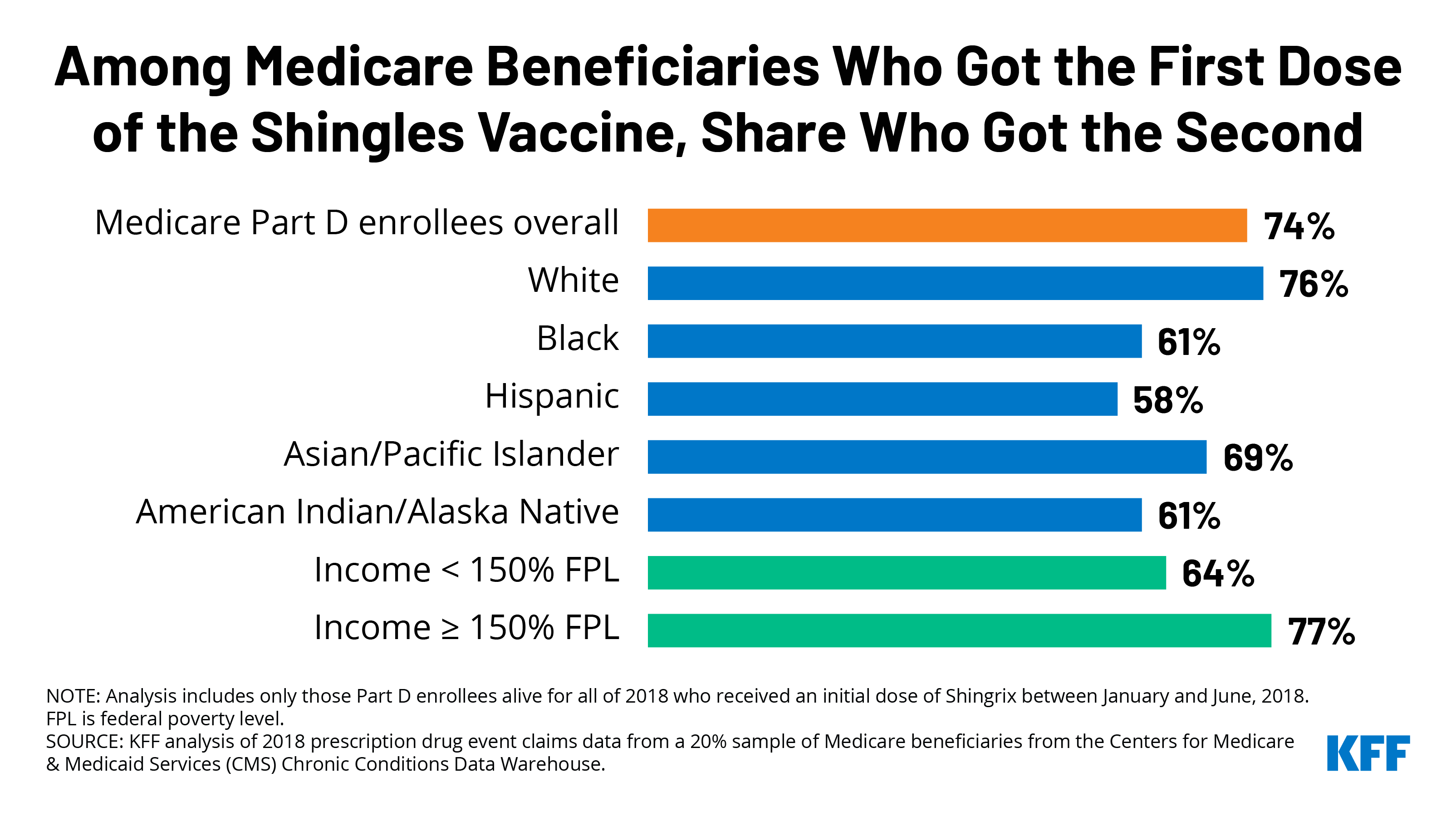
COVID-19 Vaccine Second-Dose Completion and Interval Between First and Second Doses Among Vaccinated Persons — United States, December 14, 2020−February 14, 2021 | MMWR

Comparison between one and two dose SARS-CoV-2 vaccine prioritization for a fixed number of vaccine doses | Journal of The Royal Society Interface
Phase 1/2 Study to Describe the Safety and Immunogenicity of a COVID-19 RNA Vaccine Candidate (BNT162b1) in Adults 18 to 55 Year

Public health impact of delaying second dose of BNT162b2 or mRNA-1273 covid-19 vaccine: simulation agent based modeling study | The BMJ
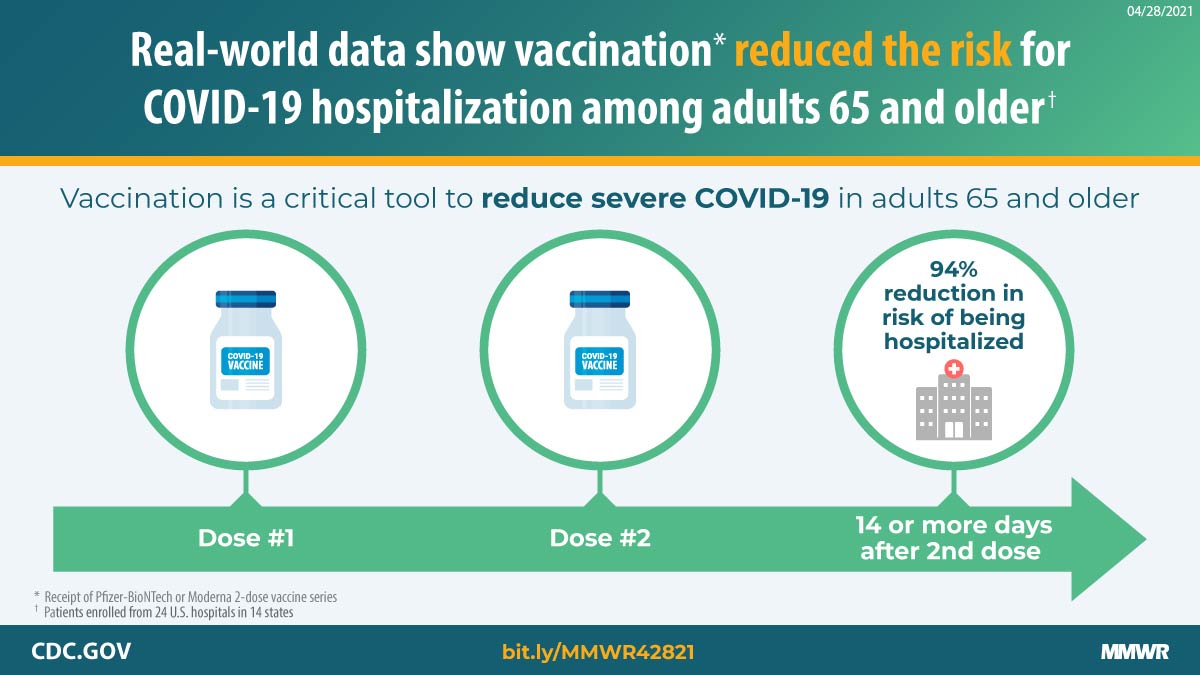
Effectiveness of Pfizer-BioNTech and Moderna Vaccines Against COVID-19 Among Hospitalized Adults Aged ≥65 Years — United States, January–March 2021 | MMWR