
Copper wire reacts with silver nitrate to form silver and copper (1) nitrate. This reaction is - Brainly.com

STOCK IMAGE, , JB7956, 01B466TS , Science Source - Search Medical & Scientific Stock Photos at MedicalImages.com

Royal Society of Chemistry on Twitter: "A Christmas tree made of crystals of pure silver, grown from copper and silver nitrate, won students from Gordon's School in Woking first prize in an

How to Balance CuSO4 + AgNO3 = Ag2SO4 + Cu(NO3)2 | Copper (II) Sulfate plus Silver Nitrate - YouTube

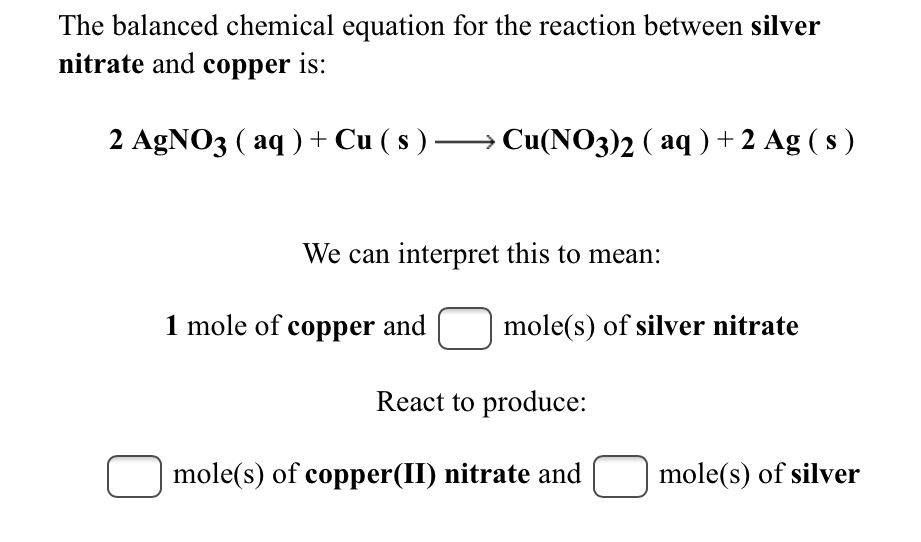
SOLVED: The reaction between solid copper and aqueous silver nitrate produces solid silver and blue solution, as represented by the balanced equation shown below: Cu(s) 2AgNO (aq) Cu(NO )(aq) 2Ag(s) Based on

SOLVED: Table 6.3 This photo shows the results of putting copper metal into a solution of silver nitrate. The progress of the reaction is shown: Copper initial Silver nitrate initial After mixing
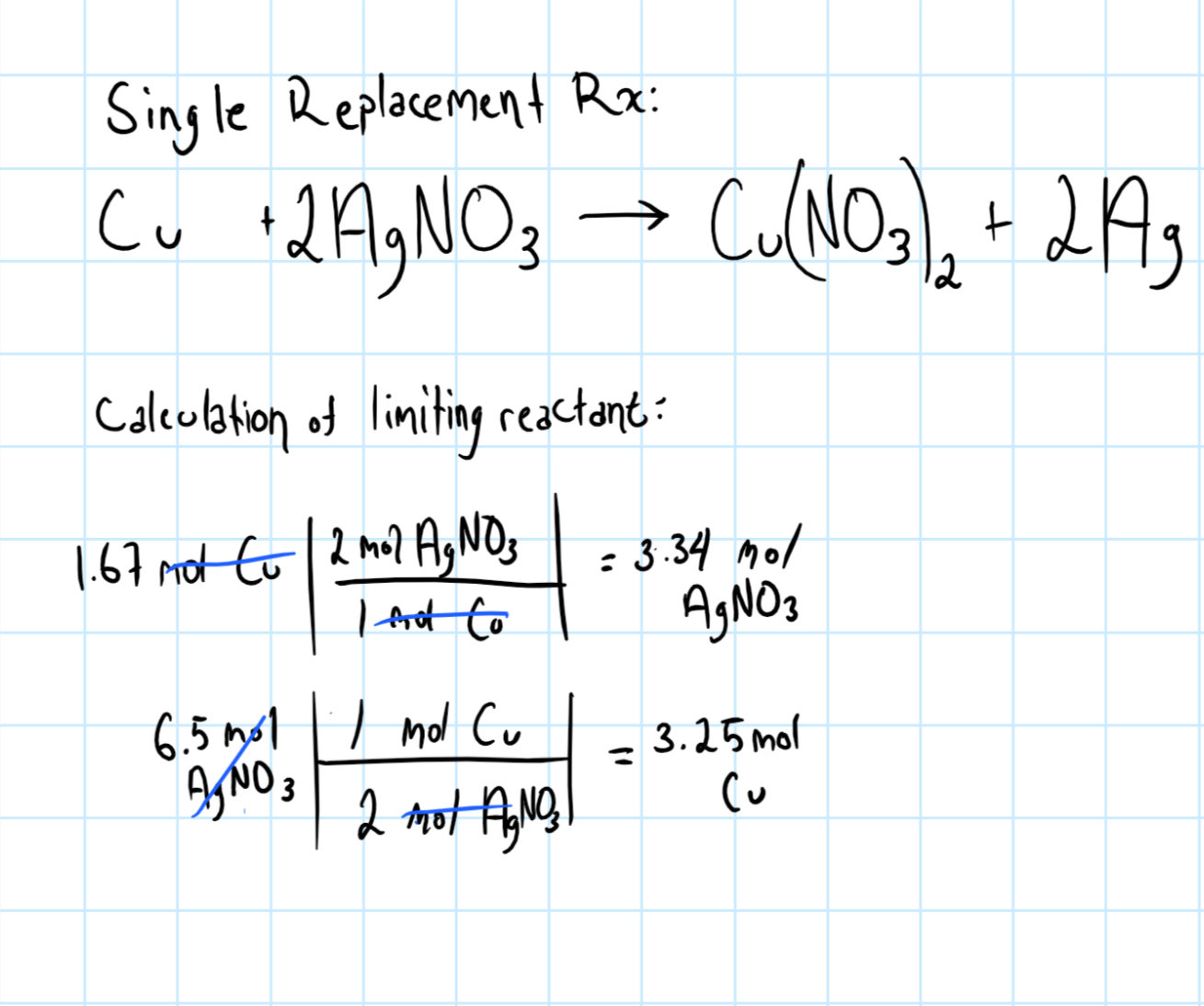












/copper-wire-immersed-in-silver-nitrate-causing-blue-colour-81991997-582f14595f9b58d5b1a9b484.jpg)




