If 34.5 g of copper reacts with 70.2 g of silver nitrate, according to the following reaction, what is the maximum number of grams of silver that can be produced? - Quora

SOLVED: Table 6.3 This photo shows the results of putting copper metal into a solution of silver nitrate. The progress of the reaction is shown: Copper initial Silver nitrate initial After mixing

Copper wire reacts with silver nitrate to form silver and copper (1) nitrate. This reaction is - Brainly.com

Observe the diagram showing a copper rod kept immersed in silver nitrate solution.a. What is the colour change of the solution?b. Write the balanced chemical equation for the reaction.
Solved] Solid copper sulfide and silver nitrate react to form copper (II) nitrate and solid silver sulfide. Write a balanced chemical equation that... | Course Hero

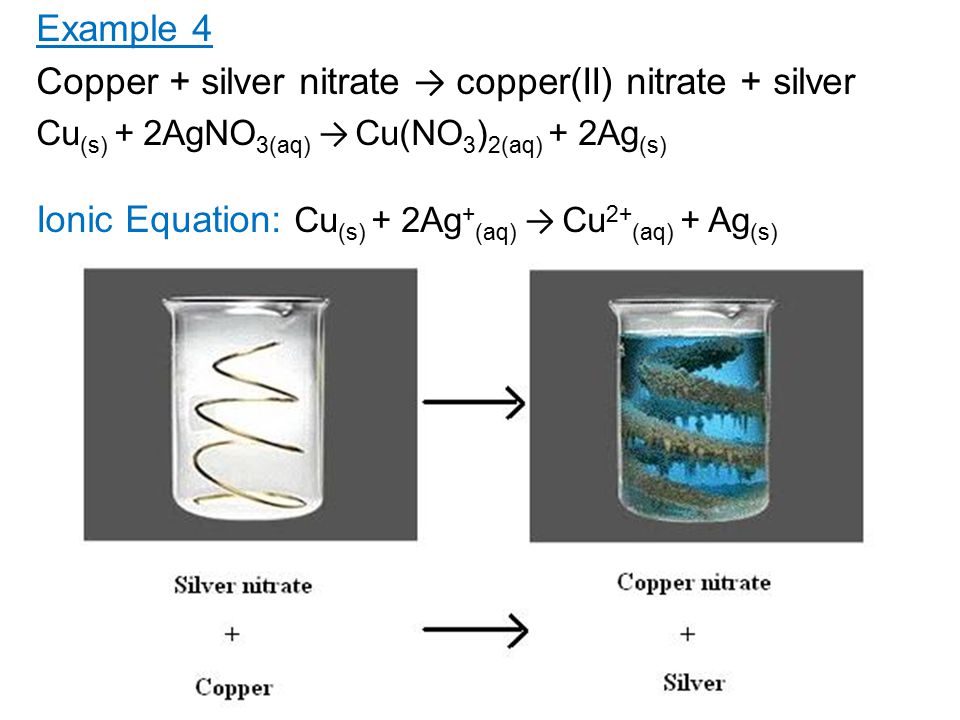

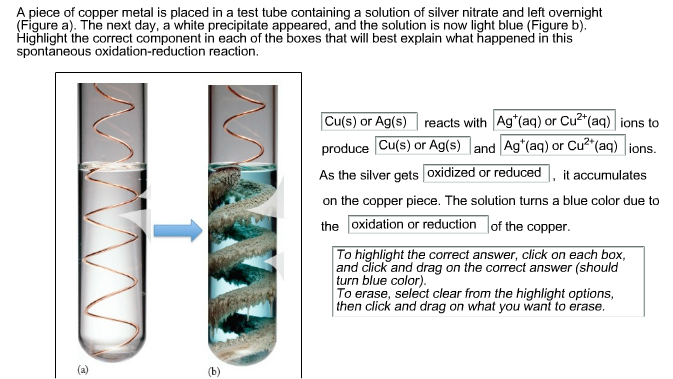







/copper-wire-immersed-in-silver-nitrate-causing-blue-colour-81991997-582f14595f9b58d5b1a9b484.jpg)








